|
|
CONTRACEPTIVE
TECHNOLOGY
FROM the
FDA
WEBSITE
(Feb. 2016)
http://www.fda.gov/forconsumers/byaudience/forwomen/freepublications/ucm313215.htm
|
|
(1) BARRIER METHODS ; (2) SPERMICIDE ALONE ; (3) HORMONAL METHODS ; (4) I.U.Ds ; (5) STERILIZATION
[Please note that the following text not only contains inaccuracies that we will discuss in class, but it promotes a mindset completely opposed to Catholic moral teaching. Nevertheless, it is important that we familiarize ourselves with this text, since it is easily available on the Internet to persons of all ages; and we must be able to answer the questions people will ask us after reading this kind of information].
| www.fda.gov/fdac/features/
1997/397_baby.html U.S. Food and Drug Administration FDA Consumer magazine April 1997 Table of Contents; 2013 POSTER |
This article originally appeared in the April 1997issue of FDA Consumer, and contains revisions made in June 1997, October 1999, and June 2000. The article is no longer up-to-date. For the most recent information on this topic, see the accompanying 2015 Women_Health_Topics. ; alt: [server] 2015 Women Health Topics |
Protecting
Against Unintended Pregnancy:
A Guide to Contraceptive Choices
by Tamar Nordenberg
I am 20 and have never gone to see a doctor about birth control. My boyfriend and I have been going together for a couple of years and have been using condoms. So far, everything is fine. Are condoms alone safe enough, or is something else safe besides the Pill?
--Letter to the Kinsey Institute for Research in Sex, Gender, and Reproduction
This young woman is not alone in her uncertainty about contraceptive options. A 1995 report by the National Academy of Sciences’ Institute of Medicine, The Best Intentions: Unintended Pregnancy and the Well-being of Children and Families, attributed the high rate of unintended pregnancies in the United States, in part, to Americans’ lack of knowledge about contraception. About six of every 10 pregnancies in the United States are unplanned, according to the report.
Being informed about the pros and cons of various contraceptives is important not only for preventing unintended pregnancies but also for reducing the risk of illness or death from sexually transmitted diseases (STDs), including AIDS. (See “Preventing HIV and Other STDs.”)
The Food and Drug Administration has approved a number of birth control methods, ranging from over-the-counter male and female condoms and vaginal spermicides to doctor-prescribed birth control pills, diaphragms, intrauterine devices (IUDs), injectable hormones, and hormonal implants. Other contraceptive options include fertility awareness and voluntary surgical sterilization.
“On the whole, the contraceptive choices that Americans have are very safe and effective,” says Dennis Barbour, former president of the Association of Reproductive Health Professionals, “but a method that is very good for one woman may be lousy for another.”
The choice of birth control depends on factors such as a person’s health, frequency of sexual activity, number of partners, and desire to have children in the future. Effectiveness rates, based on statistical estimates, are another key consideration. (See “Birth Control Guide.”) FDA has developed a consumer-friendly table of pregnancy rates, which the agency encourages all contraceptives marketers to add to their products’ labeling. Single copies of the table may be ordered from FDA, HFZ-210, 1350 Piccard Drive, Rockville, MD 20850.
(1) BARRIER METHODS
|
|
|
The male condom is a sheath placed over the erect penis before penetration, preventing pregnancy by blocking the passage of sperm.
A
condom can be used only once. Some have a chemical added to kill sperm The
addition of this spermicide, usually nonoxynol-9 in the United States, has not
been scientifically shown to provide additional contraceptive protection over
the condom alone. Because it acts as a mechanical barrier, a condom prevents
direct contact with semen, infectious genital secretions, and genital lesions
and discharges.
Most
condoms are made from latex rubber, while a small percentage are made from lamb
intestines (sometimes called “lambskin” condoms). Condoms made from a type
of plastic called polyurethane have been marketed in the United States since
1994.
Except for abstinence, latex condoms are the most effective method for reducing the risk of infection from the viruses that cause AIDS, other HIV-related illnesses, and other STDs.
|
Note,
however, that the reported 11% failure rate |
For people who are sensitive to latex, polyurethane condoms are a good alternative.
Some condoms are prelubricated. These lubricants do not increase birth control or STD protection. Non-oil-based lubricants, such as water or K-Y jelly, can be used with latex or lambskin condoms, but oil-based lubricants, such as petroleum jelly (Vaseline), lotions, or massage or baby oil, should not be used because they can weaken the condom and cause it to break.
Failure Rate (number of pregnancies expected per 100 women per year): 11 (a, b)
Some Risks (serious medical risks from contraceptives are rare): Irritation and allergic reactions (less likely with polyurethane)
Protection from Sexually Transmitted Diseases (STDs): Except for abstinence, latex condoms are the best protection against STDs, including herpes and AIDS.
Convenience: Applied immediately before intercourse; used only once and discarded. Polyurethane condoms are available for those with latex sensitivity.
Availability: Nonprescription
|
[2] FEMALE CONDOM
|
|
The
Reality Female Condom, approved by FDA in April 1993, consists of a lubricated
polyurethane sheath shaped similarly to the male condom. The closed end, which
has a flexible ring, is inserted into the vagina, while the open end remains
outside, partially covering the labia.
The female condom, like the male condom, is available without a prescription and is intended for one-time use. It should not be used together with a male condom because they may slip out of place.
Failure Rate (number of pregnancies expected per 100 women per year): 21
Some Risks (serious medical risks from contraceptives are rare): Irritation and allergic reactions
Protection from Sexually Transmitted Diseases (STDs): May give some STD protection; not as effective as latex condom
Convenience: Applied immediately before intercourse; used only once and discarded.
Availability: Nonprescription
| [3] DIAPHRAGM |  |
|
Available
by prescription only and sized by a health professional to achieve a proper fit,
the diaphragm is a dome-shaped rubber disk with a flexible rim that works in two
ways to prevent pregnancy. It covers the cervix so sperm can’t reach the
uterus, while a spermicide cream or jelly applied to the diaphragm before
insertion kills sperm.
The diaphragm protects for six hours after it is inserted. For intercourse after the six-hour period, or for repeated intercourse within this period, fresh spermicide should be placed in the vagina with the diaphragm still in place. The diaphragm should be left in place for at least six hours after the last intercourse but not for longer than a total of 24 hours because of the risk of toxic shock syndrome (TSS), a rare but potentially fatal infection. Signs and symptoms of TSS include sudden fever, stomach upset, sunburn-like rash, and a drop in blood pressure.
Failure Rate (number of pregnancies expected per 100 women per year): 17 (b, d, e)
Some Risks (serious medical risks from contraceptives are rare): Irritation and allergic reactions, urinary tract infection. (c) Risk of Toxic Shock Syndrome, a rare but serious infection, when kept in place longer than recommended.
Protection from Sexually Transmitted Diseases (STDs): None
Convenience: Inserted before intercourse and left in place at least six hours after; can be left in place for 24 hours, with additional spermicide for repeated intercourse.
Availability: Prescription
| [3] CERVICAL CAP |  |
|
The
cervical cap is a soft rubber cup with a round rim, sized by a health
professional to fit snugly around the cervix. It is available by prescription
only and, like the diaphragm, is used with spermicide cream or jelly.
It protects for 48 hours and for multiple acts of intercourse within this time. Wearing it for more than 48 hours is not recommended because of the risk, though low, of TSS. Also, with prolonged use of two or more days, the cap may cause an unpleasant vaginal odor or discharge in some women.
Failure Rate (number of pregnancies expected per 100 women per year): 17 (b, d, e)
Some Risks (serious medical risks from contraceptives are rare): Irritation and allergic reactions, abnormal Pap test. (c) Risk of Toxic Shock Syndrome, a rare but serious infection, when kept in place longer than recommended.
Protection from Sexually Transmitted Diseases (STDs): None
Convenience: May be difficult to insert; can remain in place for 48 hours without reapplying spermicide for repeated intercourse.
Availability: Prescription
| [3] SPONGE | 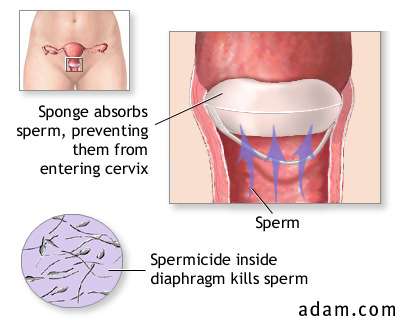 |
|
The
sponge, a disk-shaped polyurethane device containing the spermicide nonoxynol-9,
is not currently marketed but may be sold again in the future. Inserted into the
vagina to cover the cervix, the sponge is attached to a woven polyester loop for
easier removal.
The sponge protects for up to 24 hours and for multiple acts of intercourse within this time. It should be left in place for at least six hours after intercourse but should be removed no more than 30 hours after insertion because of the risk, though low, of TSS.
Failure Rate (number of pregnancies expected per 100 women per year): 14-28 (d, e)
Some Risks (serious medical risks from contraceptives are rare): Irritation and allergic reactions, difficulty in removal. (c) Risk of Toxic Shock Syndrome, a rare but serious infection, when kept in place longer than recommended.
Protection from Sexually Transmitted Diseases (STDs): None
Convenience: Inserted before intercourse and protects for repeated acts of intercourse for 24 hours without additional spermicide; must be left in place for at least six hours after intercourse; must be removed within 30 hours of insertion. Is discarded after use.
Availability: Nonprescription; not currently marketed
|
(2) VAGINAL SPERMICIDES |
|
Vaginal
spermicides are available in foam, cream, jelly, film, suppository, or tablet
forms. All types contain a sperm-killing chemical.
Studies
have not produced definitive data on how well spermicides alone prevent
pregnancy, but according to the authors of Contraceptive Technology, a leading
resource for contraceptive information, the failure rate for typical users may
be 26 percent per year.
Package instructions must be carefully followed because some spermicide products require the couple to wait 10 minutes or more after inserting the spermicide before having sex. One dose of spermicide is usually effective for one hour. For repeated intercourse, additional spermicide must be applied. And after intercourse, the spermicide has to remain in place for at least six to eight hours to ensure that all sperm are killed. The woman should not douche or rinse the vagina during this time.
Failure Rate (number of pregnancies expected per 100 women per year): 20-50 (studies have shown varying Failure Rates)
Some Risks: (serious medical risks from contraceptives are rare): Irritation and allergic reactions, urinary tract infections (c)
Protection from Sexually Transmitted Diseases (STDs): None
Convenience: Instructions vary; check labeling. Inserted between 5 and 90 minutes before intercourse and usually left in place at least six to eight hours after.
Availability: Nonprescription
(3)
HORMONAL METHODS
|
All hormonal methods are probably abortifacient in an unknown percentage of cases, through their action of preventing normal development of the uterine endometrium (lining of the womb), and thus preventing implantation of the newly-fertilized embryo. |
|
[1] COMBNED ORAL CONTRACEPTIVES |
|
Typically called “the pill,” combined oral contraceptives have been on the market for 40 years and are the most popular form of reversible birth control in the United States. This form of birth control suppresses ovulation (the monthly release of an egg from the ovaries) by the combined actions of the hormones estrogen and progestin.
If a woman remembers to take the pill every day at the same time of day as directed, she has an extremely low chance of becoming pregnant. But the pill’s effectiveness may be reduced if the woman is taking some medications, such as certain antibiotics.
Besides preventing pregnancy, the pill offers additional benefits. As stated in the labeling, the pill can make periods more regular and lighter. It also has a protective effect against pelvic inflammatory disease, an infection of the fallopian tubes or uterus that is a major cause of infertility in women, and against ovarian and endometrial cancers.
|
This is "one-sided reporting" at it's worst! The "periods" a women experiences while on the pill are not normal menstruation. And the risk of PID is obviously dramatically increased by frequent intercourse with different partners. |
The decision whether to take the pill should be made in consultation with a health professional. Birth control pills are safe for most women--safer even than delivering a baby--but they carry some risks.
Current low-dose pills have fewer risks associated with them than earlier versions. But women over age 35 who smoke and women with certain medical conditions, such as a history of blood clots or breast or endometrial cancer, may be advised against taking the pill. The pill may contribute to cardiovascular disease, including high blood pressure, blood clots, and blockage of the arteries.
One of the biggest questions has been whether the pill increases the risk of breast cancer in past and current pill users. An international study published in the September 1996 journal Contraception concluded that women’s risk of breast cancer 10 years after going off birth control pills was no higher than that of women who had never used the pill. During pill use and for the first 10 years after stopping the pill, women’s risk of breast cancer was only slightly higher in pill users than non-pill users Women who have or have had breast cancer should not use the pill because the estrogen in the pill may worse their medical condition.
Side effects of the pill, which often subside after a few months’ use, include nausea, headache, breast tenderness, weight gain, irregular bleeding, and depression.
Failure Rate (number of pregnancies expected per 100 women per year): 1
Some Risks (serious medical risks from contraceptives are rare): Dizziness; nausea; changes in menstruation, mood, and weight; rarely, cardiovascular disease, including high blood pressure, blood clots, heart attack, and strokes
Protection from Sexually Transmitted Diseases (STDs): None, except some protection against pelvic inflammatory disease
Convenience: Must be taken on daily schedule, regardless of frequency of intercourse.
Availability: Prescription
[2]
MINIPILLS.
Although
taken daily like combined oral contraceptives, minipills contain only the
hormone progestin and no estrogen. They work by reducing and thickening cervical
mucus to prevent sperm from reaching the egg. They also
keep the uterine lining from thickening, which prevents a fertilized egg from
implanting in the uterus. [i.e. THIS
METHOD IS PRIMARILY ABORTIFICIANT] These pills are slightly less
effective than combined oral contraceptives.
Minipills,
like combined oral contraceptives, can decrease menstrual bleeding and cramps
and lower the risk of endometrial and ovarian cancer and pelvic inflammatory
disease. Because they contain no estrogen, minipills don’t present the risk of
blood clots associated with estrogen in combined pills. They are a good option
for new mothers who are breast-feeding, because combined oral contraceptives may
decrease the quantity and quality of breast milk. They are also a good option
for those who get severe headaches or high blood pressure from
estrogen-containing products.
Side
effects of minipills include menstrual cycle changes, weight gain, and breast
tenderness.
|
[3] EMERGENCY CONTRACEPTIVE (“Morning After Pill”) [= ABORTIFICIANT] |
|
Two emergency contraceptive pill products have been approved by FDA for use in preventing pregnancy after intercourse when standard contraceptives have failed or when no contraceptives were used at all. One product contains the hormones progestin and estrogen; the other contains just progestin.
Available by prescription only, both products are believed to work by delaying or inhibiting ovulation, or by keeping a fertilized egg from implanting in the uterine wall. These pills are not effective once the fertilized egg has implanted.
Emergency contraceptives are about 75 percent effective, which means the number of women who would be expected to become pregnant after unprotected sex drops from eight without the “morning after pill” to two when it is used.
Side effects include nausea and vomiting, both of which were reported less frequently in women taking the progestin-only pills.
Failure Rate (number of pregnancies expected per 100 women per year): Almost 80 percent reduction in risk of pregnancy for a single act of unprotected sex [i.e. FAILURE = 20%]
Some Risks (serious medical risks from contraceptives are rare): Nausea, vomiting, abdominal pain, fatigue, headache
Protection from Sexually Transmitted Diseases (STDs): None
Convenience: Must be taken within 72 hours of having unprotected intercourse.
Availability: Prescription
|
[4] INJECTABLE PROGESTINS |
|
Depo-Provera,
approved by FDA in 1992, is injected by a health professional into the buttocks
or arm muscle every three months. Depo-Provera prevents pregnancy in three ways:
It inhibits ovulation, changes the cervical mucus to help prevent sperm from
reaching the egg, and changes the uterine lining to
prevent the fertilized egg from implanting in the uterus. The
progestin injection is extremely effective in preventing pregnancy, in large
part because it requires little effort for the woman to comply: She simply has
to get an injection by a doctor once every three months.
The benefits are similar to those of the minipill and another progestin-only contraceptive, Norplant. Side effects are also similar and can include irregular or missed periods (which is not harmful and does not mean that the method isn’t working), weight gain, and breast tenderness.
Failure Rate (number of pregnancies expected per 100 women per year): less than 1
Some Risks (serious medical risks from contraceptives are rare): Irregular bleeding, weight gain, breast tenderness, headaches
Protection from Sexually Transmitted Diseases (STDs): None
Convenience: One injection every three months.
Availability: Prescription
|
[5] IPLANTABLE PROGESTINS |
|
Norplant, approved by FDA in 1990, and the newer Norplant 2, approved in 1996, are the third type of progestin-only contraceptive. Made up of matchstick-sized rubber rods, this contraceptive is surgically implanted under the skin of the upper arm, where it steadily releases the contraceptive steroid levonorgestrel.
The
six-rod Norplant provides protection for up to five years (or until it is
removed), while the two-rod Norplant 2 protects for up to three years. Norplant
failures are rare, but are higher with increased body weight.
Some women may experience inflammation or infection at the site of the implant. Other side effects include menstrual cycle changes, weight gain, and breast tenderness.
Failure Rate (number of pregnancies expected per 100 women per year): less than 1
Some Risks (serious medical risks from contraceptives are rare): Irregular bleeding, weight gain, breast tenderness, headaches, difficulty in removal
Protection from Sexually Transmitted Diseases (STDs): None
Convenience: Implanted by health-care provider in minor outpatient surgical procedure; effective for up to five years.
Availability: Prescription. In July 2002, Norplant’s manufacturer announced that it will no longer distribute the Norplant system. Women using the system should contact their doctors about what their contraceptive options will be after the five-year expiration date of their Norplant systems.
(4) INTRAUTERINE DEVICES
 |
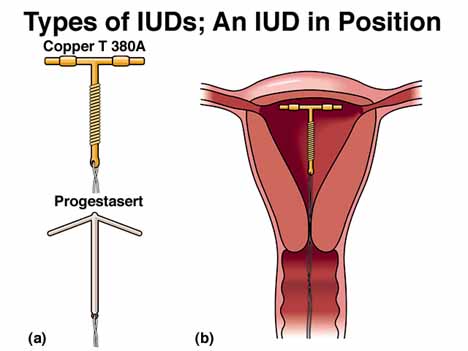 |
An
IUD is a mechanical device inserted into the uterus by a health-care
professional. Two types of IUDs are available in the United States: the Paragard
CopperT 380A and the Progestasert Progesterone T. The Paragard IUD can remain in
place for 10 years, while the Progestasert IUD must be replaced every year.
It’s
not entirely clear how IUDs prevent pregnancy. They seem to prevent sperm and
eggs from meeting by either immobilizing the sperm on their way to the fallopian
tubes or changing the uterine lining so the fertilized
egg cannot implant in it.
IUDs have one of the lowest failure rates of any contraceptive method. “In the population for which the IUD is appropriate--for those in a mutually monogamous, stable relationship who aren’t at a high risk of infection--the IUD is a very safe and very effective method of contraception,” says Lisa Rarick, M.D., former director of FDA’s division of reproductive and urologic drug products.
The IUD’s image suffered when the Dalkon Shield IUD was taken off the market in 1975. This IUD was associated with a high incidence of pelvic infections and infertility, and some deaths. Today, serious complications from IUDs are rare. Side effects can include pelvic inflammatory disease (an infection of a woman’s reproductive organs), ectopic pregnancy (in which a fertilized egg implants in the fallopian tube instead of the uterus), perforation of the uterus, heavier-than-normal bleeding, and cramps. Complications occur most often during and immediately after insertion.Failure Rate (number of pregnancies expected per 100 women per year): less than 1
Some Risks (serious medical risks from contraceptives are rare): Cramps, bleeding, pelvic inflammatory disease, infertility, perforation of uterus
Protection from Sexually Transmitted Diseases (STDs): None
Convenience: After insertion by physician, can remain in place for up to one or 10 years, depending on type.
Availability: Prescription
(5) STERILIZATION
|
|
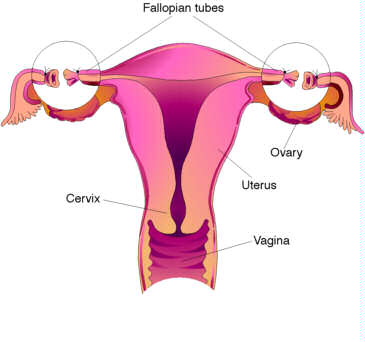 |
Surgical sterilization is a contraceptive option intended for
people who don't want children in the future. It is considered permanent because
reversal requires major surgery that is often unsuccessful.
Female sterilization: Tubal Ligation
Female sterilization blocks the fallopian tubes so the egg
can't travel to the uterus. Sterilization is done by various surgical
techniques, usually under general anesthesia.
Complications from these operations are rare and can include infection, ectopic pregnancy, hemorrhage, and problems related to the use of general anesthesia.
The woman's fallopian tubes are blocked so the egg and sperm
can't meet in the fallopian tube, preventing conception. (g)
Failure Rate (number of pregnancies expected per 100 women per year): less than
1
Some Risks (serious medical risks from contraceptives are rare): Pain, bleeding,
infection, other post-surgical complications
Protection from Sexually Transmitted Diseases (STDs): None
Convenience: One-time surgical procedure.
Availability: Surgery
Male sterilization: Vasectomy.
This procedure, called a vasectomy, involves sealing, tying or
cutting a man's vas deferens, which otherwise would carry the sperm from the
testicle to the penis.
Failure Rate (number of pregnancies expected per 100 women per
year): less than 1
Some Risks (serious medical risks from contraceptives are rare): Pain, bleeding,
infection, other minor postsurgical complications
Protection from Sexually Transmitted Diseases (STDs): None
Convenience: One-time surgical procedure.
Availability: Surgery
Medieval Obstetrical Instruments
xxxx» cont
xcxcxc